

CASE STUDY
Empowered Innovation:
People Science and Mimio Recreate Fasting Benefits with a Novel Fasting Mimetic
Study Overview
Primary focus
Evaluate the impact of the Mimio™ fasting mimetic on daily hunger and satiety.
Participants
42 participants, 47.6% women.
Study Design
Double-blind, placebo-controlled, randomized, decentralized trial approved by the Advarra Institutional Review Board (IRB).
Study Structure
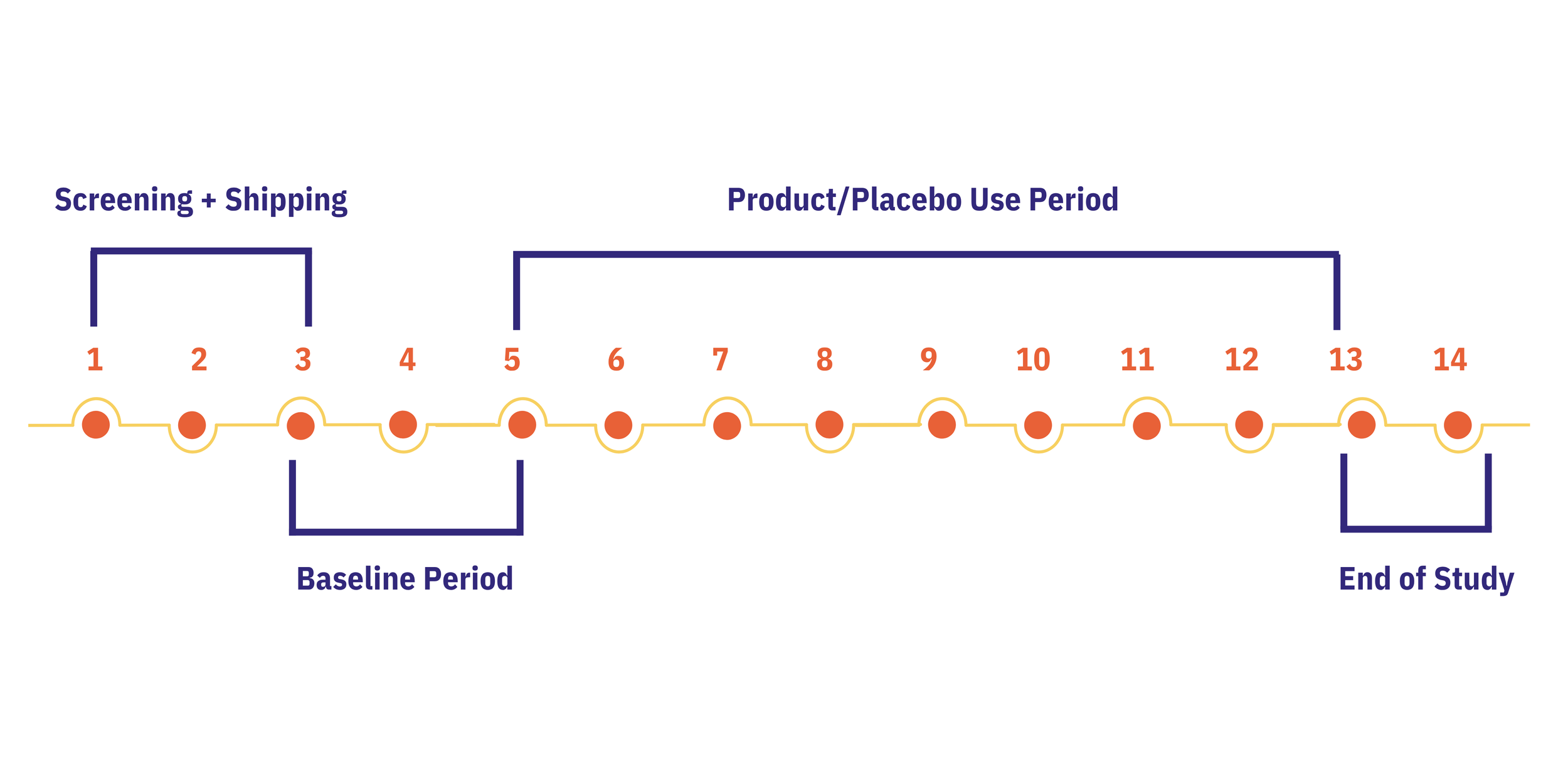
Participants collected 2 weeks of baseline data followed by 8 weeks of product or placebo use. The study was completely decentralized, with all outcomes logged via the Chloe app.
Duration
14-week study including screening and shipping period, 2-week baseline period, and 8-week product or placebo use period.
Data Collection
Metabolic bloodwork, daily scale of hunger & satiety, digestive symptoms and quality of life, cognitive failures questionnaire, eating behavior questionnaire.
Publication
A manuscript describing the findings is published at Nature Scientific Reports and available here, DOI: 10.1038/s41598-026-38495-7.
Study Snapshot
Key Results
91%
Decreased Mealtime Appetite
87.6%
Eliminated Bloating
8.6%
Drop in Oxidized LDL, an Oxidative Stress Marker
83.3%
Eliminated Abdominal Pain
94%
Adherence
Study Details

Introduction
Prolonged fasting is one of the few interventions consistently shown to extend lifespan and health span in model organisms. Despite its robust potential in clinical applications to treat, prevent, or delay many major diseases, the safety concerns, impracticality and patient burden associated with fasting limit widespread adoption.
Mimio Health, an innovative team studying biomimetics, launched an initiative to validate the efficacy of its novel fasting mimetic “Mimio™”— intended to curb hunger, increase satiety and support metabolic health. Its formulation consists of spermidine, nicotinamide, palmitoylethanolamide (PEA), and oleoylethanolamide (OEA), which are small molecules produced naturally by the human body during extended fasts. In a previous pilot clinical study, these metabolites were well-tolerated and provided anti-inflammatory, antioxidant, and heart-protective benefits similar to fasting.
To further the understanding of fasting mimetics and demonstrate product efficacy, Mimio Health partnered with People Science. This collaboration gathered concrete scientific evidence for how this combination of molecules generates many functional benefits of prolonged fasting—without the need to fast.

Execution
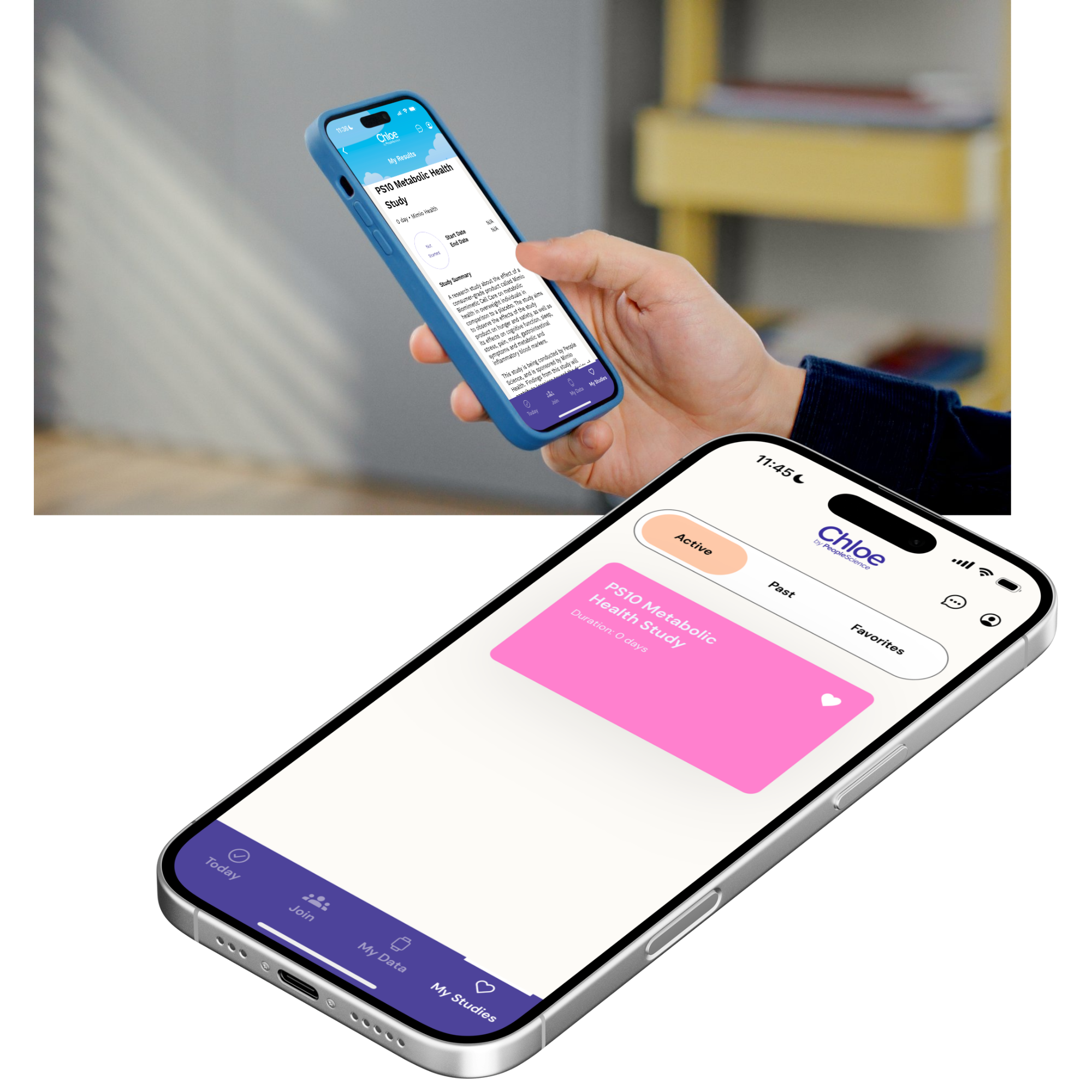
To execute a decentralized, double-blind, placebo-controlled study, People Science utilized its proprietary Consumer Health Learning & Organizing Ecosystem (Chloe). Chloe combines a mobile app with a health research platform designed for companies that prioritize scientific rigor, enabling rapid and cost-effective validation of product claims through evidence-based methods.
Decentralized Clinical Trials (DCTs) evaluate the impact of novel interventions without the need for in-person doctor’s visits. By combining a mobile app for communication, consent, data entry and visualization, a software backend for clinical operations and a team to manage shipments - all the work of participating in a clinical trial can be done from home. This strategy is speeding every stage of scientific advancement, from cost reduction to study design, recruitment, data collection, monitoring, and data visualization.

Data Collection
The cohort consisted of overweight adults aged 55 and older with elevated HbA1c (6.0%+). Participants were considered evaluable for analysis if they completed 8 weeks of Mimio/placebo use, baseline measurements and at least 70% of their questionnaires. Blood was drawn after at least an overnight fast at baseline and end of the study for evaluation of key blood markers, including glucose, insulin, HbA1c, and NMR lipid panel.
During the 8-week product or placebo use period, participants were asked to consume one capsule daily (prior to their first major meal of the day). After each day, participants completed a custom Hunger, Satiety and Cravings Scale consisting of 7 items on a 0-5 scale. Each week, participants also completed questionnaires on digestive symptoms and quality of life, eating behavior and the cognitive failures questionnaire (CFQ). 42 participants were evaluated (47.6% female and 52.4% male).
The double-blind design ensured that neither the participants nor the researchers knew which type of capsules were consumed, prioritizing the scientific integrity of the study.
Outcome
Clinical Validation
The study found that Mimio™ supplementation over 8 weeks reduced hunger and boosted satiety. One of the most striking metrics was reduction in appetite: 91% reported improved appetite vs. 47% for placebo. These results are statistically significant, relevant and will contribute both to the scientific understanding of fasting mimetics and to product development.
Decentralized Trial Efficiency
The Chloe platform was instrumental in the Mimio Health trial, providing an end-to-end solution for decentralized data collection and seamless participant engagement. All assessments and surveys were administered in the Chloe app, significantly reducing logistical complexity for both the trial sponsor and participants.
Effective Execution
Completed the trial over a 14-week period. This project demonstrated People Science’s full-service support capabilities, from study design to data analysis.

“People Science allowed us to conduct a rigorous, gold-standard trial directly in participants' homes. The data we gathered not only confirmed our mechanism of action but showed that our formulation can deliver the benefits of fasting to the population that needs it most.”
Dr. Chris Rhodes, PhD
CEO of Mimio Health

Benefits to Mimio
-
This study is the first to show that fasting mimetic supplementation can recreate clinical fasting-like cardiometabolic benefits without lifestyle changes or food restriction. These results position Mimio as an innovative leader in the health and wellness industry.
Mimio’s ability to reduce LDL and total cholesterol, oxidized LDL and glucose, alongside substantial improvements in hunger, satiety, and digestive comfort, sets it apart from conventional supplements and positions it as a disruptive innovation in cardiometabolic health. By offering a practical and tolerable alternative to fasting regimens, Mimio expands access to fasting-related health benefits for individuals unable or unwilling to fast. These clinical findings not only validate Mimio’s health optimization claims but also create opportunities for expanded research, product development, and industry partnerships. Trial results are published in Nature Scientific Reports (DOI: 10.1038/s41598-026-38495-7).
-
People Science is revolutionizing the landscape of clinical trials by making them more accessible and affordable for consumer health, wellness, device and nutrition brands. By integrating advanced technology with clinical expertise, the company redesigns the evaluation process to deliver precise measurement, deeper understanding, and more impactful health outcomes.
-
Provided instant insights into participant data, allowing timely adjustments to maximize study effectiveness.
-
Data accuracy is essential for reliable clinical trials, ensuring that every measurement and data point reflects true outcomes without error or bias. Structured prompts and reminders helped maintain high data quality and participant adherence throughout the trial.

Benefits to
Trial Participants
-
Allowed participants to track their experiences with Mimio™ from home or on the go, enhancing accessibility.
-
Participants received personalized results in the app at the end of the study, as well as the “first look” at overall trial results. Expanding the participant role beyond data collection to data interpretation improves personal utility of joining a study as well as data literacy.
-
Chloe dashboards and reports kept collaborators up-to-date with study progress.
-
The platform’s usability scored 4.5/5.

I appreciate the text reminders. The daily survey only took a few minutes. Response to questions through the app was great. Helpful and courteous staff—happy to help through participation in future studies!
Study Participant

Book a Demo
Turn your customers’ everyday health data into your most powerful competitive asset. Explore our Chloe platform today.



